AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Pi bonds vs sigma bonds1/17/2024  
Therefore, we can conclude that NCO does not have a linear structure. It is a polar molecule with a dipole moment resulting from the asymmetric distribution of electrons around the nitrogen and carbon atoms. NCO, on the other hand, has a bent or angular shape with a bond angle that is not 180 degrees. This is because it is isoelectronic with CO2, which is a linear molecule. However, it is important to note that the closely related −NCO (−N=C=O) molecule is linear. The NCO- molecule has a unique structure due to its ambidentate ligand nature and linear geometry. 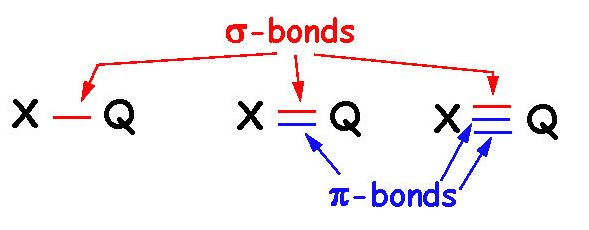
The nitrogen atom is bonded to the carbon atom by a triple bond, and the carbon atom is bonded to the oxygen atom by a double bond. The NCO- molecule is made up of three atoms: nitrogen, carbon, and oxygen. It is an ambidentate ligand, whch means that it can donate electrons from either the nitrogen or oxygen site. The NCO- molecule has a negative charge present over the nitrogen and oxygen atoms due to electron resonance. The linear geometry of the molecule results in a bond angle of 180 degrees. NCO- is an anion that has a linear geometry with sp hybridization. The NCO- molecule is a chemical compound that has a unique structure. It is important to note that the Lewis structure does not account for the molecular geometry of NCO, which is linear. The triple bond betwen the carbon and nitrogen atoms is composed of one sigma bond and two pi bonds. In this structure, the nitrogen atom has a lone pair of electrons and a formal charge of -1, while the carbon atom has a formal charge of +1 and the oxygen atom has a formal charge of -1. The carbon atom is also bonded to the oxygen atom. The nitrogen atom is the central atom and is bonded to both the carbon and oxygen atoms. 
The Lewis structure for NCO, also known as isocyanate, is composed of one nitrogen atom, one carbon atom, and one oxygen atom. Understanding the molecule’s resonance structure is key to understanding its properties and reactivity. Its ambident nucleophile properties make it incredibly useful in a wide range of chemical reactions. The NCO- molecule is a complex and versatile molecule that has a unique Lewis structure. It is important to be aware of thee similarities and differences, as they can impact how the molecule behaves in different chemical reactions. One example is NCO+, which is a non-linear molecule. It is worth noting that there are other molecules that are similar to NCO. This resonance structure is important because it helps to explain some of the molecule’s unique properties. This results in the carbon atom displaying positive charge, while the nitrogen and oxygen atoms display negative charge simultaneously. In terms of its resonance structure, the carbon atom in the NCO- molecule is induced by the nitrogen and oxygen atoms. This property makes the molecule incredibly versatile in terms of its reactivity. As such, it is known as an ambidentate ligand. This is because it has the ability to donate electrons from either the nitrogen atom or the oxygen atom. The NCO- molecule is regarded as an ambident nucleophile in nucleophilic substitution reactions. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
